Spectroscopy and Astronomy
 Astronomers generally do not look through their large telescopes.
Most of the time large telescopes are collecting light for a
spectrograph, which spreads the light out into a rainbow. Each kind of
atom or ion has certain special wavelengths which it can absorb or emit.
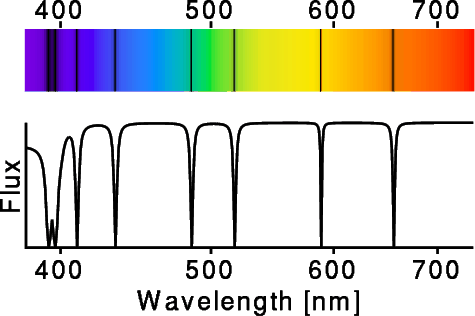
Atoms in the cool outer layers of a star absorb light coming from the
hotter regions within, producing dark absorption lines across the
spectrum. These can be used to identify the atoms that make up a star.
In the spectrum at right, the dark lines are 2 from ionized calcium
at about 390 nm
wavelength, hydrogen lines at 410, 434, 486 and 656 nm,
a line from ionized magnesium at 518 nm, and a line from sodium at 590
nm.
To see the lines produced by any element, use this
Java applet.
Astronomers generally do not look through their large telescopes.
Most of the time large telescopes are collecting light for a
spectrograph, which spreads the light out into a rainbow. Each kind of
atom or ion has certain special wavelengths which it can absorb or emit.
Atoms in the cool outer layers of a star absorb light coming from the
hotter regions within, producing dark absorption lines across the
spectrum. These can be used to identify the atoms that make up a star.
In the spectrum at right, the dark lines are 2 from ionized calcium
at about 390 nm
wavelength, hydrogen lines at 410, 434, 486 and 656 nm,
a line from ionized magnesium at 518 nm, and a line from sodium at 590
nm.
To see the lines produced by any element, use this
Java applet.
And astronomers do not actually use color film to take a color picture
of the spectrum, for several reasons:
- Color film or CCDs are less sensitive than black and white.
- Color film does a very poor job of representing the continuous range
of colors in a spectrum. It takes a lot of work to make a decent
printout or display of a spectrum.
- While a picture may be worth a thousand words, a graph showing
brightness vs. wavelength has much more quantitative
information than a picture of the spectrum.
Thus astronomers make graphs of their spectra, with the y-axis showing
the brightness, while the x-axis shows the wavelength. In the figure
this graph is aligned just below the picture of the spectrum.
Tutorial:
Part 1 |
Part 2 |
Part 3 |
Part 4
FAQ |
Age |
Distances |
Bibliography |
Relativity
Ned Wright's home page
© 1999 Edward
L. Wright. Last modified 28-Aug-1999
 Astronomers generally do not look through their large telescopes.
Most of the time large telescopes are collecting light for a
spectrograph, which spreads the light out into a rainbow. Each kind of
atom or ion has certain special wavelengths which it can absorb or emit.
Atoms in the cool outer layers of a star absorb light coming from the
hotter regions within, producing dark absorption lines across the
spectrum. These can be used to identify the atoms that make up a star.
In the spectrum at right, the dark lines are 2 from ionized calcium
at about 390 nm
wavelength, hydrogen lines at 410, 434, 486 and 656 nm,
a line from ionized magnesium at 518 nm, and a line from sodium at 590
nm.
To see the lines produced by any element, use this
Java applet.
Astronomers generally do not look through their large telescopes.
Most of the time large telescopes are collecting light for a
spectrograph, which spreads the light out into a rainbow. Each kind of
atom or ion has certain special wavelengths which it can absorb or emit.
Atoms in the cool outer layers of a star absorb light coming from the
hotter regions within, producing dark absorption lines across the
spectrum. These can be used to identify the atoms that make up a star.
In the spectrum at right, the dark lines are 2 from ionized calcium
at about 390 nm
wavelength, hydrogen lines at 410, 434, 486 and 656 nm,
a line from ionized magnesium at 518 nm, and a line from sodium at 590
nm.
To see the lines produced by any element, use this
Java applet.